News&Event
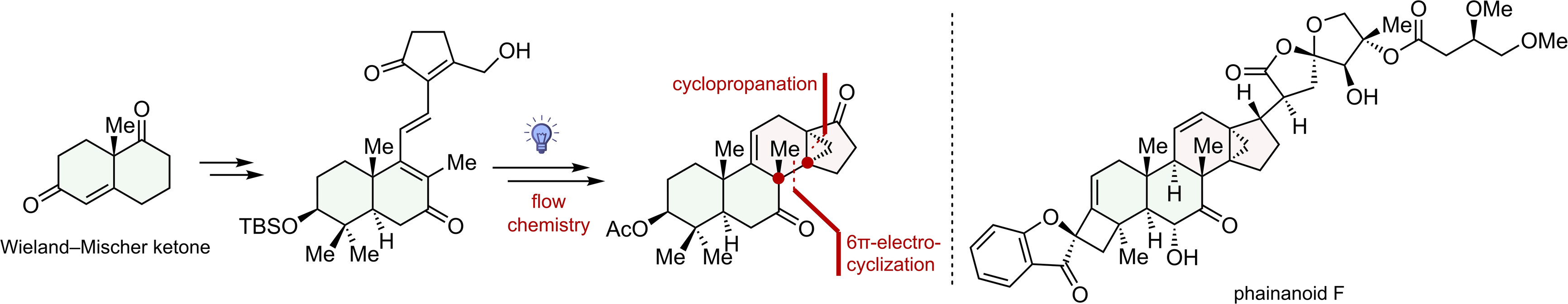
170.Synthesis towards Phainanoid F: Photo-induced 6π- Electrocyclization for Constructing Contiguous All-Carbon Quaternary Centers
Hao-Yuan Liu, Zhen-Yu Zhang, Yi-Ke Zhou, Jia-Hua Chen, Zhen Yang,* and Yuan-He Li*
Chem Asian J.2023,18, e20230062
In this paper, we report an efficient strategy for synthesizing the DEFGH rings of phainanoid F. The key to the construction of the 13,30-cyclodammarane skeleton of the molecule was a photo-induced 6π-electrocyclization and a homoallylic elimination. Notably, this is a rare example of using electrocyclization reaction to simultaneously construct two vicinal quaternary carbons in total synthesis. The strategy outlined here forms the basis of our total synthesis of Phainanoid F, and it could also serve as a generally applicable approach for synthesizing other natural products containing similar 13,30-cyclodammarane skeletons.
169.Regioselective Hydroxylation of Flavonoids by Transition-Metal-Catalyzed C−H Bond Oxidation
Shu-Min Lu, Chao Chen, Chang Liu, Rudong Liu, Jia-Hua Chen,* Zhongchao Zhang,* and Zhen Yang*
Org. Lett. 2023, 25(13), 2264–2269
Regioselective synthesis of 5,6,7-trihydroxyl and 5,7,8-trihydroxyl flavones has been achieved via a transition-metal-catalyzed C–H oxidation as the key step using naturally enriched 5,7-dihydroxyl flavone. The developed chemistry was applied to the synthesis of the naturally occurring and biologically active flavonoids wogonin (2), oroxylin A (3), and their glycosylated derivatives (4 and 5) as potential carnitine palmitoyltransferase 1 activators.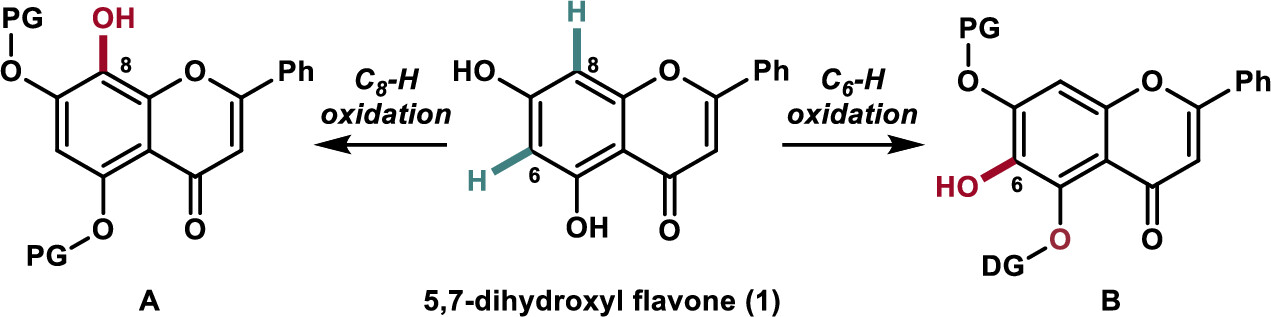
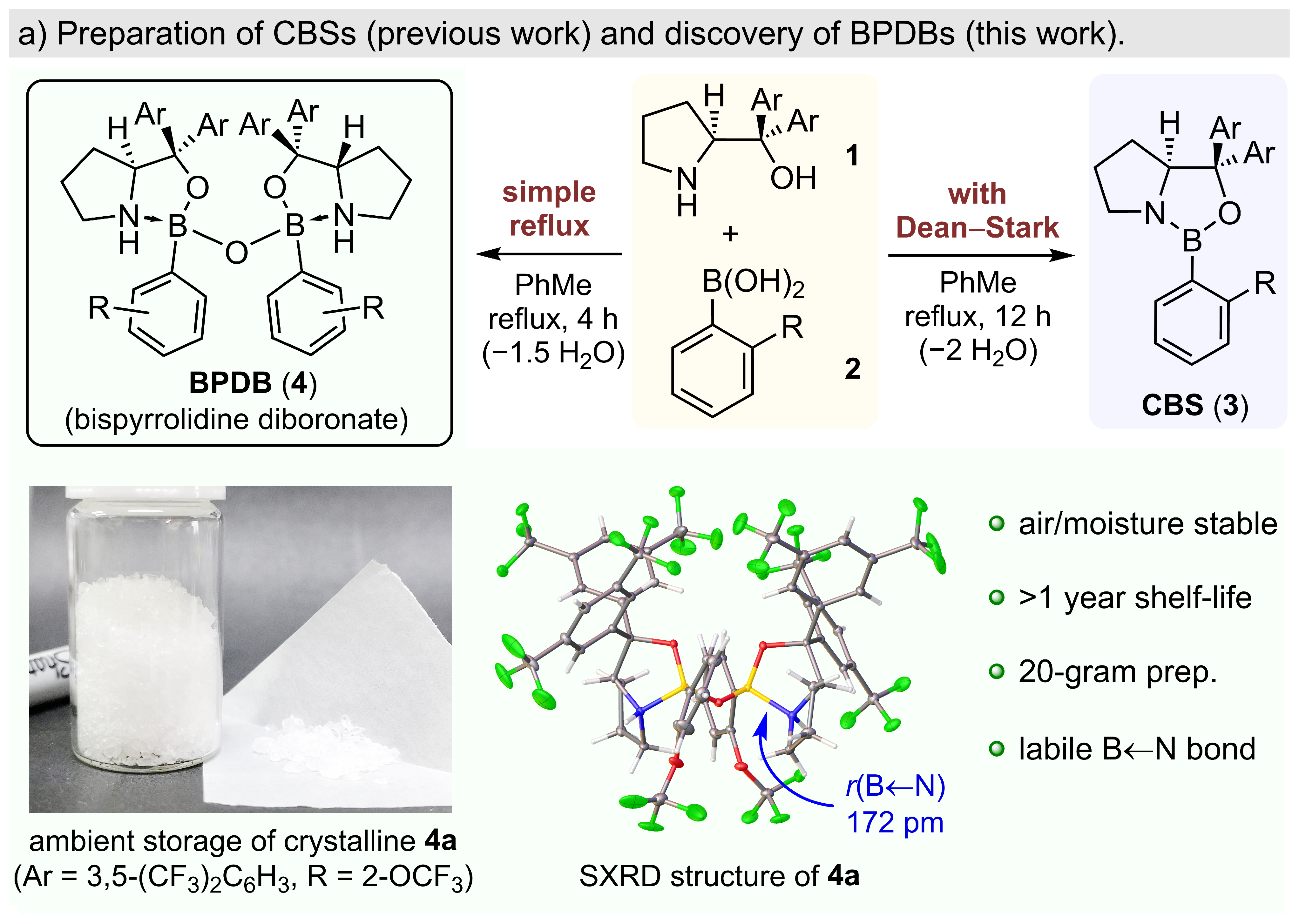
168.Highly Stereoselective Diels-Alder Reactions Catalyzed by Diboronate Complexes
Yuan-He Li, Su-Lei Zhang, Yong Lu, Bo Xiao, Tian-Yu Sun, Qian-Qian Xu, Jia-Hua Chen,* Zhen Yang*
Angew. Chem. Int. Ed. 2023, 62(33)
A highly enantioselective catalytic system for exo-Diels–Alder reactions was developed based on the newly discovered bispyrrolidine diboronates (BPDBs). When activated various Lewis or Brønsted acids, BPDBs could catalyze highly stereoselective asymmetric exo-Diels–Alder reactions of monocarbonyl-based dienophiles. When 1,2-dicarbonyl-based dienophiles are used, the catalyst can sterically distinguish between the two binding sites, which leads to highly regioselective asymmetric Diels–Alder reactions. BPDBs can be prepared as crystalline solids on a large scale and are stable under ambient conditions. Single-crystal X-ray analysis of the structure for acid-activated BPDB indicated that activation involves breakage of a labile N→B bond.
167.Synthesis of the 5/6 fused-spiro tricyclic framework of manginoid A
Zhiming Yan, Chunbo Zhao, Zhen Yang*, Jianxian Gong*
Tetrahedron Letters 2023, 117 (2023), 154361-154363
We developed a concise, ten-step route to the 5/6 fused-spiro tricyclic framework (A/B/C rings) of manginoid A from the starting materials cyclopentenone and 1,3-cyclohexanedione. Michael addition and subsequent Mukaiyama aldol reaction introduced two side chains at the α,β-positions of cyclopentenone. Tsuji–Trost allylation of this cyclopentenone moiety and 1,3-cyclohexanedione afforded a key intermediate. A late-stage Michael addition formed the spiro-fused tricyclic framework.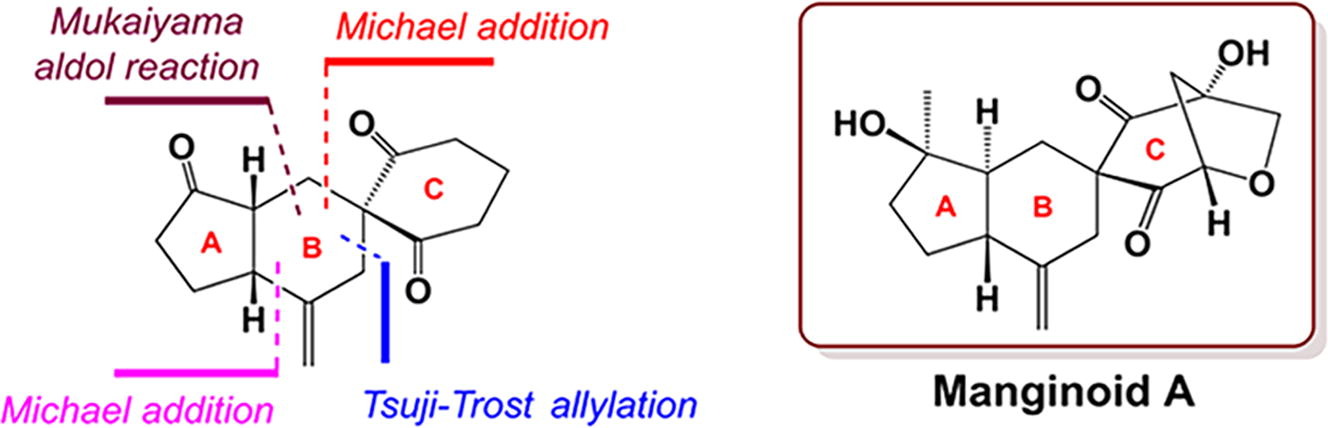
166.Recent advances in total synthesis of natrual products containing contiguous all carbon quaternary stereocenters
Zhongchao Zhang, Zhen Yang *
SCIENTIA SINICA Chimica 2023, 53(3), 277-288
Contiguous all carbon quaternary stereocenters are common structural units in bioactive natural products with significant synthetic challenges. In this paper, we reviewed our recent progresses in total synthesis of complex natural products containing contiguous all carbon quaternary stereocenters firstly. Then, four different synthetic strategies concerning the construction of contiguous all carbon quaternary stereocenters (≥3) were summarized. Finally, the total synthesis of waihoensene and crinipellins reported by us and other groups were taken as examples to clarify the advantages and disadvantages of various synthetic methodologies and synthetic strategies.