Congratulations to Feng Xiong for his paper published on Nature Communication
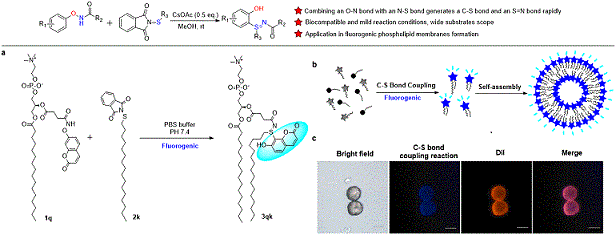
Synthetic methods inspired by Nature often offer unique advantages including mild conditions and biocompatibility with aqueous media. Inspired by an ergothioneine biosynthesis protein EgtB, a mononuclear non-haem iron enzyme capable of catalysing the C–S bond formation and sulfoxidation, herein, we discovered a mild and metal-free C–H sulfenylation/intramolecular rearrangement cascade reaction employing an internally oxidizing O–N bond as a directing group. Our strategy accommodates a variety of oxyamines with good site selectivity and intrinsic oxidative properties. Combining an O–N bond with an X–S bond generates a C–S bond and an S ¼ N bond rapidly. The newly discovered cascade reaction showed excellent chemoselectivity and a wide substrate scope for both oxyamines and sulfenylation reagents. We demonstrated the biocompatibility of the C–S bond coupling reaction by applying a coumarin-based fluorogenic probe in bacterial lysates. Finally, the C–S bond coupling reaction enabled the first fluorogenic formation of phospholipids, which self-assembled to fluorescent vesicles in situ.