Xijian Li’s work has been published in Adv. Synth. Catal.!
Amine-Triggered 6p-Electrocyclization–Aromatization Cascade of Ynedienamines
Xijian Li, Huidong Yu, and Yong Huang*, Adv. Synth. Catal. 2017, 359, 1379 – 1387
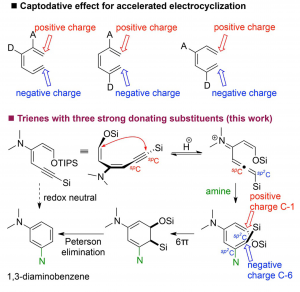
An unprecedented 6p-electrocyclization–aromatization cascade reaction of ynedienamines is described. The electron-rich ynedienamine is converted by g-protonation to an electrophilic allenyl iminium species which is susceptible to amine addition generating a highly electron-rich triene intermediate. The 6p-electrocyclization is accelerated by three electron-donating substituents in a captodative manner. Subsequent redox-neutral aromatization allows the direct synthesis of 1,3-diaminobenzenes from readily available ynedienamines.