Jiean Chen and Pengfei Yuan’s paper has been published in J. Am. Chem. Soc.! Congratulations!
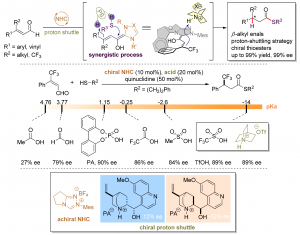
Enantioselective β‑Protonation of Enals via a Shuttling Strategy
Jiean Chen, Pengfei Yuan, Leming Wang, and Yong Huang*, J. Am. Chem. Soc. 2017, 139, 7045−7051
Remote asymmetric protonation is a longstanding challenge due to the small size of protons. Reactions involving electron-deficient olefins pose a further difficulty due to the electrophilic nature of these substrates. We report a shuttling system that delivers a proton in a highly enantioselective manner to the β-carbon of enals using a chiral N-heterocyclic carbene (NHC) catalyst. Choices of a Brønsted base shuttle and a Brønsted acid cocatalyst are critical for highly stereoselective β-protonation of the homoenolate intermediate and regeneration of the NHC catalyst results in functionalization of the carbonyl group. Thioesters with a β-chiral center were prepared in a redox-neutral transformation with an excellent yield and ee.