News&Event
Congratulation to Yun Zhang for her review paper was published on NPR!
Congratulation to Yun Zhang for her paper has been published on Natural Product Reports! This review titled “Strategic innovation in the total synthesis of complex natural products using gold catalysis”. For more details, please see the paper online:
https://web.pkusz.edu.cn/yang/79/
or
Nat. Prod. Rep., 2014, 31, 489
Zhang’s page: https://web.pkusz.edu.cn/yang/zhangy/
80.Strategic innovation in the total synthesis of complex natural products using gold catalysis
Yun Zhang, Tuoping Luo* and Zhen Yang*
Nat. Prod. Rep., 2014, 31, 489
Novel organic reactions drive the advance of chemical synthesis in the same way that enabling technologies drive new scientific discoveries. One area of organic methodology that has undergone significant growth during the last decade is that of homogeneous gold-catalyzed transformations. This trend has been further enhanced by the employment of gold catalysis on a routine basis to accomplish the total synthesis of natural products. In particular, the superior π acidity of the cationic gold complex for the activation of alkynes and allenes towards nucleophilic addition has significantly enriched the toolkit of transformations available to the total synthesis community, and inspired a new era of creativity in terms of the strategic disconnection of target compounds during their retrosynthetic analysis. Instead of simply supplementing the many existing reviews of gold catalysis, this review has been organized from the perspective of synthetic target families, with particular emphasis on the use of gold-catalyzed transformations during the late stages of syntheses involving complicated substrates, and cascade reactions that significantly increase molecular complexity.
Congratulation to Guozong Yue for his work was published on Angew!
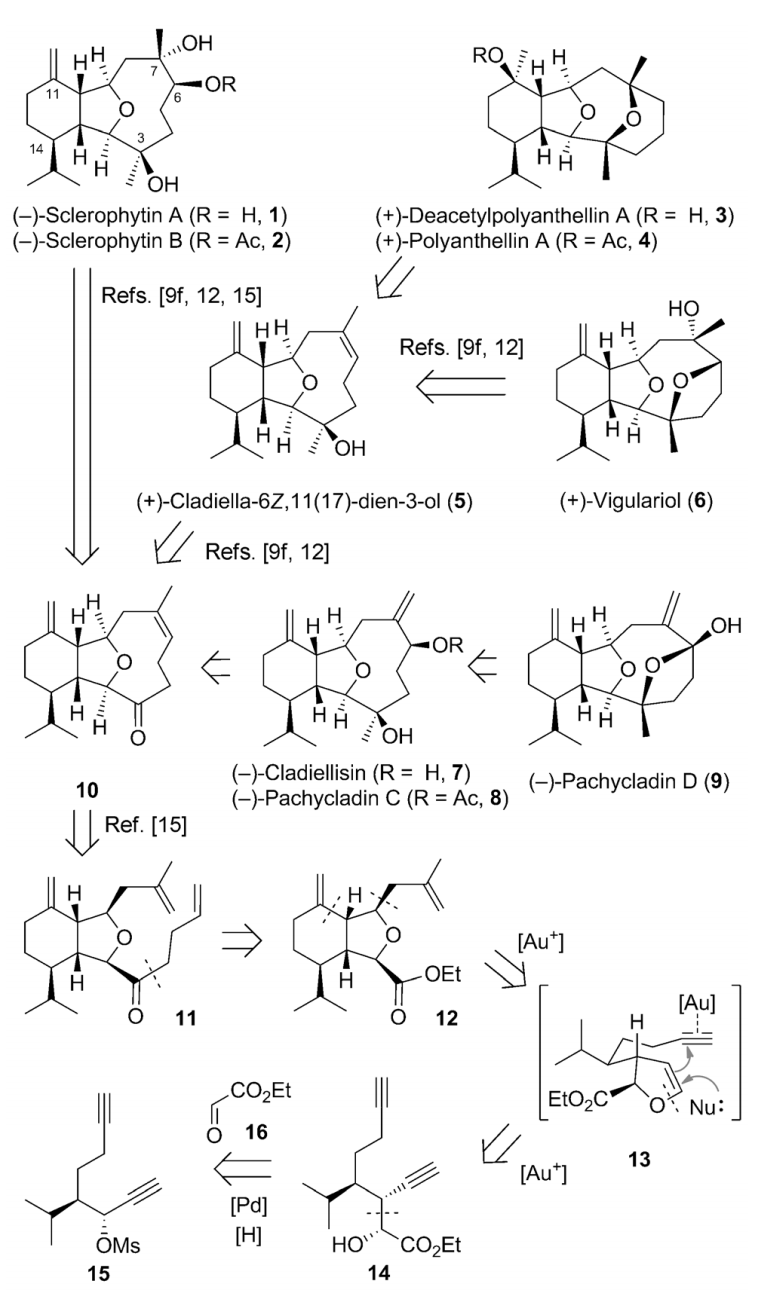
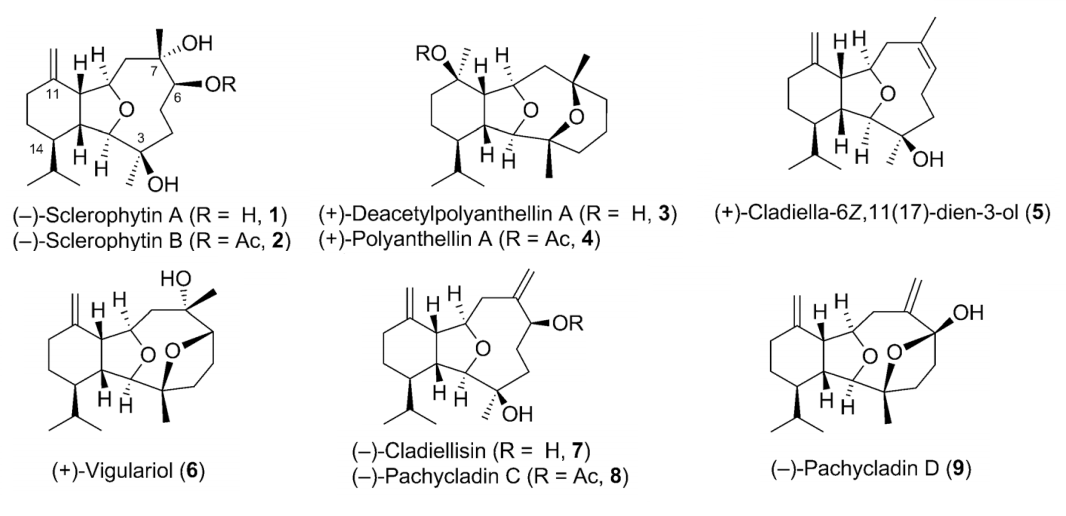
Congratulation to Guozong Yue for his work was published on Angew! He has finished the synthesis of NINE molecules in the cladiellin family:
For more details, please see the paper online:
https://web.pkusz.edu.cn/yang/78/
or
Angew. Chem. Int. Ed. 2014, 53, 1837
79.Collective Synthesis of Cladiellins Based on the Gold-Catalyzed Cascade Reaction of 1,7-Diynes
Guozong Yue, Yun Zhang, Lichao Fang, Chuang-chuang Li, Tuoping Luo,* and Zhen Yang*
Angew. Chem. Int. Ed. 2014, 53, 1837
The cladiellin family of natural products, which includes molecules with various biological activities, continues to invite new synthetic studies. A gold-catalyzed tandem reaction of 1,7-diynes to construct the 6-5-bicyclic ring systems that are present in a number of natural products was developed. This reaction was applied as the key step to realize the formal and total syntheses of nine members of the cladiellin family in an enantio- and diastereoselective manner. This modular and efficient approach could also be used for the construction of other cladiellins, as well as their analogues, for follow-up studies.