News&Event
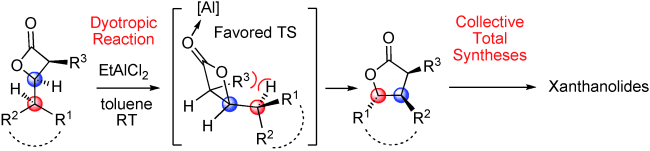
63.Enantioselective and Collective Syntheses of Xanthanolides Involving a Controllable Dyotropic Rearrangement of cis-b -Lactones
W. W. Ren, Y. C. Bian, Z. Y. Zhang, H. Shang, P. T. Zhang, Y. J. Chen, Z. Yang*, T. P. Luo*, and Y. F. Tang*
Angew. Chem. Int. Ed. 2012, 51, 6984
Let’s swap: A scalable, atom-economic, enantio-, and diastereoselective synthetic route to trisubstituted γ-butyrolactones based on a Wagner–Meerwein-type dyotropic rearrangement of cis-β-lactones is described (see scheme). This methodology was applied in efficient and protecting-group-free formal syntheses and total syntheses of various xanthanolide natural products.
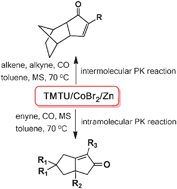
62.CoBr2-TMTU-Zinc Catalysed-Pauson-Khand Reaction
Y. F. Wang, L. M. Xu, R. C. Yu, J. H. Chen*, Z. Yang*
A cobalt–TMTU complex, derived from the in situ reduction of CoBr2 with Zn in the presence of TMTU, can catalyze Pauson–Khand reaction at a balloon pressure of CO, which enables the synthesis of structurally diverse cyclopentenones. This catalytic system works efficiently for both intermolecular and intramolecular PK reactions.
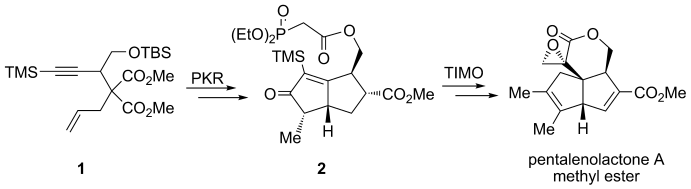
61.Total Synthesis of (±)-Pentalenolactone A Methyl Ester
Q. Liu, G. Z. Yue, N. Wu, G. Lin, Y. Z . Li, J. M. Quan, C. C. Li*, G. X. Wang*, Z. Yang*
Angew. Chem. Int. Ed. 2012, 51, 48, 12072
• Top 10 Most Accessed Article in 10/2012.
• Highlighted in Chin. J. Org. Chem., January, 2013.
• Highlighted in Chemistry World, November, 2012.
Ringing it up: The methyl ester of pentalenolactone A has been obtained through the stereoselective synthesis of a cyclopentenone by a combination of the Co-mediated Pauson–Khand reaction (PKR) of enyne 1, and the construction of a quaternary-carbon-based strained α-methylidene-δ-pentyrolactone core through a trimethylsilyl (TMS) mediated, telescoped intramolecular Michael olefination (TIMO) reaction of keto-phosphonate 2.
60.Reverse Regioselectivity in the Palladium(II) Thiourea Catalyzed Intermolecular Pauson–Khand Reaction
N. Wu, L. J. Deng, L. Z. Liu, Q. Liu, C. C. Li*, Z. Yang*
Which way? Palladium thiourea catalyzed Pauson–Khand reaction of norbornene with substituted alkynoates to afford selectively either α-carboxylated adducts or β-carboxylated adducts with the reverse regioselectivity has been demonstrated for the first time. Control of the regioselectivity in this transformation is governed by the addition of LiCl. A mechanistic interpretation to account for this observed regioselectivity is presented.
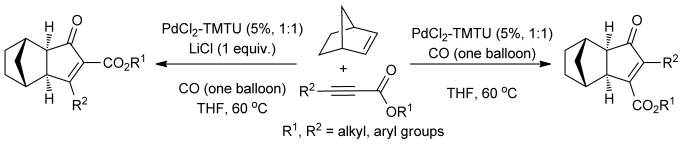
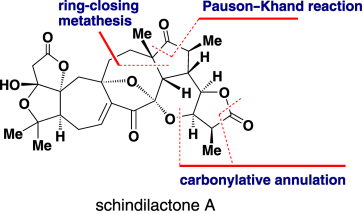
59.Diastereoselective Total Synthesis of (±)-Schindilactone A
Q. Xiao, W. W. Ren, Z. X. Chen, T. W. Sun, Y. Li, Q. D. Ye, J. X. Gong, F. K. Meng, L. You, Y. F. Liu, M. Z. Zhao, L. M. Xu, Z. H. Shan, Y. Shi,Y. F. Tang*, J. H. Chen*, Z. Yang*
Angew. Chem. Int. Ed. 2011, 50, 7373.
All together: A concise strategy for the first diastereoselective total synthesis of (±)-schindilactone A is reported. The synthesis features a ring-closing metathesis, a thiourea/cobalt-catalyzed Pauson–Khand reaction, and a thiourea/palladium-catalyzed carbonylative annulation reaction. The chemistry can be applied to the synthesis of structures related to schindilactone A.